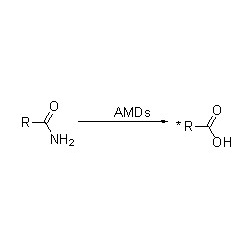
I-Amidase (AMD)
Ama-enzyme:Ziyizinto ezibangela ukwanda kwama-macromolecular biological, ama-enzyme amaningi angamaprotheni
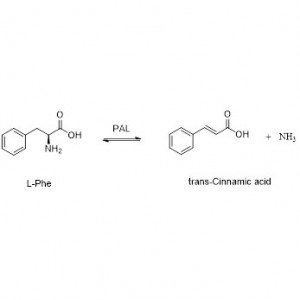
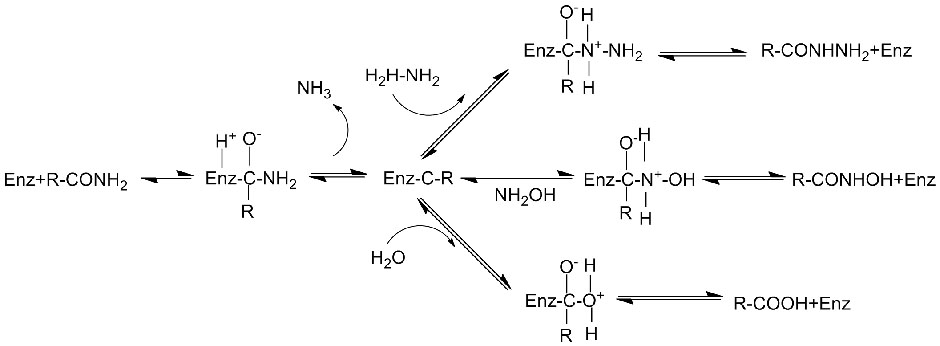
I-Amidase:Hlanganisa i-hydrolysis yama-amide ahlukahlukene e-aliphatic kanye ne-aromatic amide angaphandle nawangaphandle ngokudlulisela iqembu le-acyl emanzini ngokukhiqizwa kwama-asidi amahhala kanye ne-ammonia. Ama-asidi e-hydroxamic kanye namanye ama-asidi e-organic asetshenziswa kabanzi njengemithi ngoba ayizinto ezibangela ukukhula, ama-antibiotic kanye nezithiyo zomdlavuza. Ama-amidases angahlukaniswa abe ama-acylase ohlobo lwe-R kanye nohlobo lwe-S ngokuya nge-catalyst stereoselectivity.
Ngaphezu kokuvuselela i-hydrolysis yama-amides, i-amidase ingavuselela futhi ukusabela kokudluliselwa kwe-acyl lapho kukhona ama-co-substrate afana ne-hydroxylamine.
Ama-Amidase anemithombo ehlukene anokucaciswa okuhlukile kwe-substrate, amanye awo anga-hydrolyze ama-aromatic amides kuphela, amanye anga-hydrolyze ama-aliphatic amides kuphela, kanti amanye anga-hydrolyze ama-α-noma ama-ω-amino amides. Iningi lama-amine linomsebenzi omuhle we-catalytic kuphela kuma-amides a-acyclic noma alula e-aromatic, kodwa kuma-aromatic ayinkimbinkimbi, ama-amides e-heterocyclic, ikakhulukazi ama-amides ane-ortho substituents, ngokuvamile anomsebenzi ophansi (ama-enzyme ambalwa kuphela abonisa imiphumela engcono ye-catalytic).
Indlela yokwenza izinto nge-catalytic:
| Ama-enzyme | Ikhodi Yomkhiqizo | Ikhodi Yomkhiqizo |
| Impuphu ye-Enzyme | I-ES-AMD-101~ ES-AMD-119 | isethi yama-amidase ayi-19, ama-50 mg ngalinye izinto eziyi-19 * 50mg / into, noma olunye ubuningi |
| Ikhithi Yokuhlola (i-SynKit) | I-ES-AMD-1900 | isethi yama-amidase ayi-19, 1 mg ngayinye Izinto eziyi-19 * 1mg / into |
★ Ukucaciswa okuphezulu kwe-substrate.
★ Ukukhetha okunamandla kwe-chiral.
★ Ukusebenza kahle kokuguqulwa.
★ Imikhiqizo embalwa engaphansi.
★ Izimo zokusabela okuncane.
★ Inobungani nemvelo.
➢ Ukuhlolwa kwama-enzyme kufanele kwenziwe kuma-substrate athile ngenxa yobukhulu be-substrate, bese kutholwa i-enzyme evuselela i-substrate eqondiwe ngomphumela omuhle kakhulu we-catalytic.
➢ Ungalokothi uthinte izimo ezimbi kakhulu ezifana nalezi: izinga lokushisa eliphezulu, i-pH ephezulu/ephansi kanye nesinyibilikisi se-organic esinomthamo ophezulu.
➢ Ngokuvamile, uhlelo lokusabela kufanele lufake i-substrate, isixazululo se-buffer (I-pH yokusabela okuhle kakhulu kwe-enzyme). Ama-co-substrate afana ne-hydroxylamine kufanele abe khona ohlelweni lokusabela lwe-acyl transfer.
➢ I-AMD kufanele ifakwe ekugcineni ohlelweni lokusabela olune-pH kanye nokushisa okuphezulu kokusabela.
➢ Zonke izinhlobo ze-AMD zinezimo ezahlukene zokusabela ezingcono kakhulu, ngakho-ke ngayinye yazo kufanele ifundwe kabanzi ngayinye.
Isibonelo 1(1):
Umsebenzi we-Hydrolysis kuma-substrate ahlukene e-Amide
| I-substrate | Umsebenzi othize μmols min-1mg-1 | I-substrate | Umsebenzi othize μmols min-1mg-1 |
| I-Acetamide | 3.8 | ο-OH benzamide | 1.4 |
| I-Propionamide | 3.9 | p-OH benzamide | 1.2 |
| I-Lactamide | 12.8 | ο-NH2i-benzamide | 1.0 |
| I-Butyramide | 11.9 | p-NH2i-benzamide | 0.8 |
| I-Isobutyramide | 26.2 | ο-Toluamide | 0.3 |
| I-Pentanamide | 22.0 | p-Toluamide | 8.1 |
| I-Hexanamide | 6.4 | I-Nicotinamide | 1.7 |
| I-Cyclohexanamide | 19.5 | I-Isonicotinamide | 1.8 |
| I-Acrylamide | 10.2 | I-Picolinamide | 2.1 |
| I-Metacrylamide | 3.5 | 3-Phenylpropionamide | 7.6 |
| I-Prolinamide | 3.4 | I-Indol-3-acetamide | 1.9 |
| I-Benzamide | 6.8 |
Ukusabela kwenziwe ngesisombululo se-sodium phosphate buffer esingu-50mM, i-pH 7.5, ku-70 ℃.
| Ama-Amide | I-Hydroxylamine | I-Hydrazine |
| I-Acetamide | 8.4 | 1.4 |
| I-Propionamide | 18.4 | 3.0 |
| I-Isobutyramide | 25.0 | 22.7 |
| I-Benzamide | 9.2 | 6.1 |
Ukusabela kwenziwe ngesisombululo se-sodium phosphate buffer esingu-50mM, i-pH 7.5, ku-70 ℃.
Ukuhlushwa kwe-reagent okuhlobene: ama-amides, 100 mM(benzamide, 10 mM); i-hydroxylamine kanye ne-hydrazine, 400 mM; i-enzyme 0.9 μM.
Isibonelo 2(2):
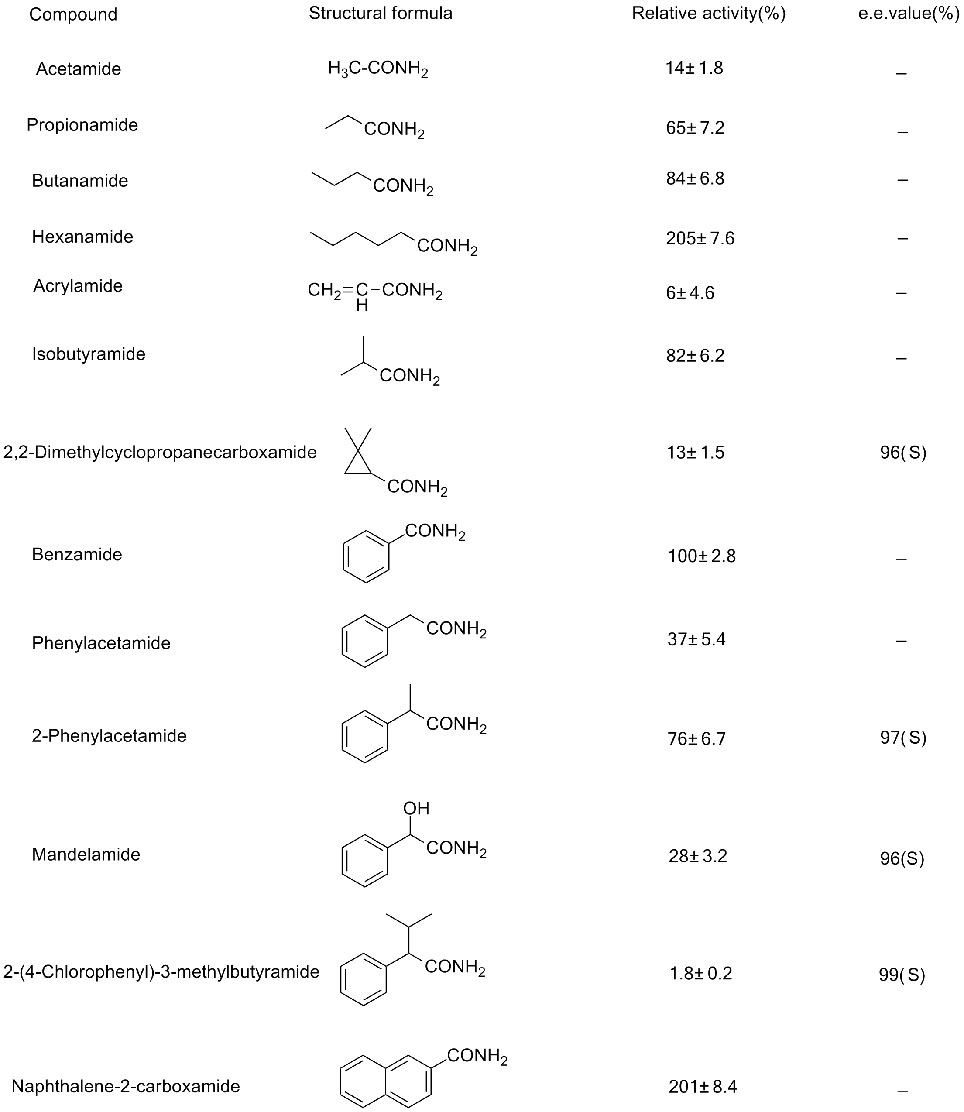
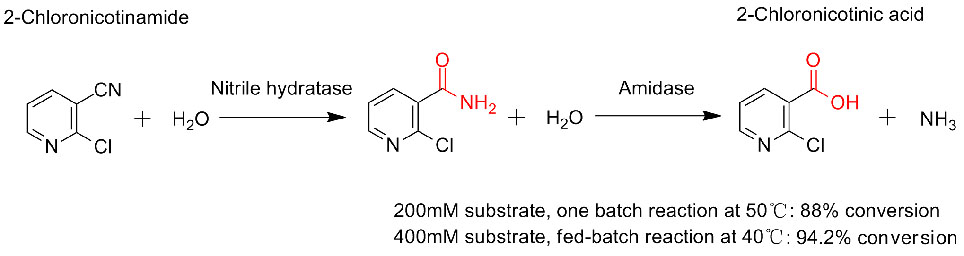
Isibonelo 3(3):
1. U-D'Abusco AS, u-Ammendola S., et al. I-Extremophiles, 2001, 5:183-192.
2. Guo FM, Wu JP, Yang LR, nabanye. I-Process Biochemistry, 2015, 50(8): 1400-1404.
3. Zheng RC, Jin JQ, Wu ZM, nabanye. I-Bioorganic Chemistry, 2017, Iyatholakala ku-inthanethi 7.